This webpage was produced as an assignment for Genetics 564, an undergraduate capstone course at the University of Wisconsin-Madison.
Post-translational modifications
|
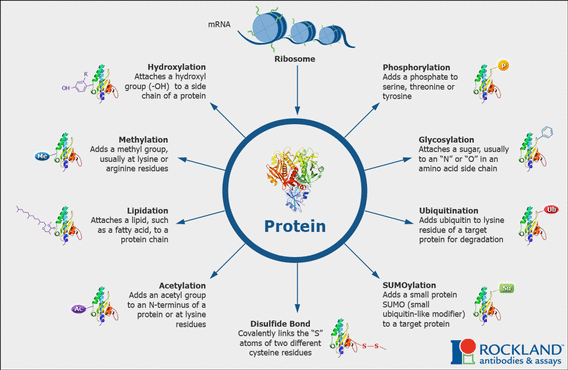
Post-translational protein modifications are often important to protein function. There are many different types of post-translational modifications, which can affect localization, folding, enzymatic activity, and more. Phosphoproteomics - the study of the phosphorylation of the proteome, in particular, is important because phosphorylation acts as a "light switch" for protein interactions and is commonly used to regulate protein activity. This may be because phosphate is a large, stable, negatively-charged, and water-soluble molecule [1]. These characteristics mean that phosphorylated amino acids can stabilize salt bridges and hydrogen bonds. The amino acids serine, thereonine, and tyrosine are most-likely to be phosphorylated in eukaryotes. Post-translational modifications can be studied by mass spectroscopy, where the wide array of modifications can be sensitively directed. However, it is important to combine in vitro and in vivo information to ensure that phosphorylation does actually occur in the protein's natural environment. |
Discussion
|
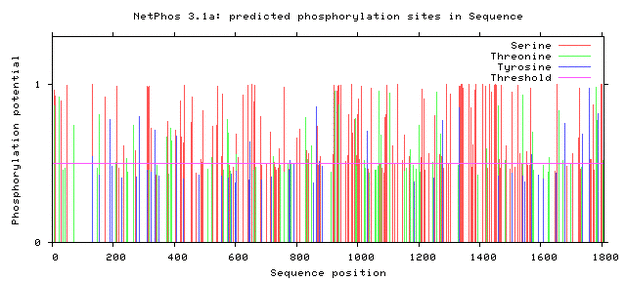
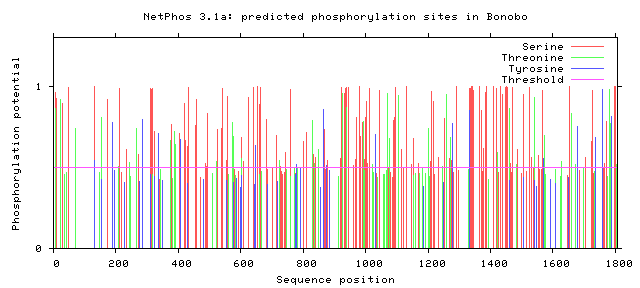
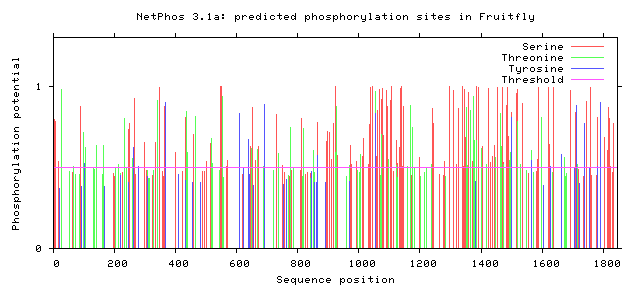
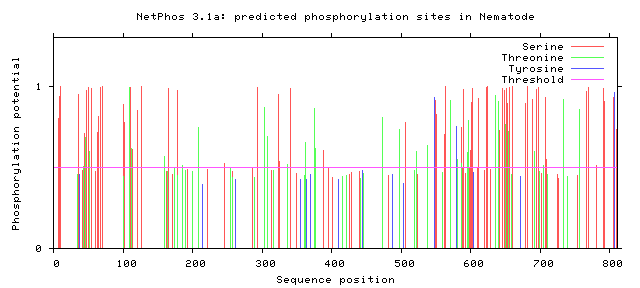
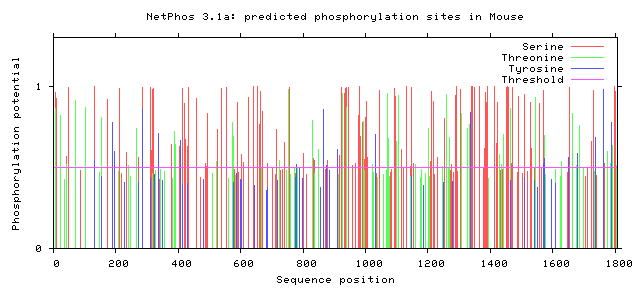
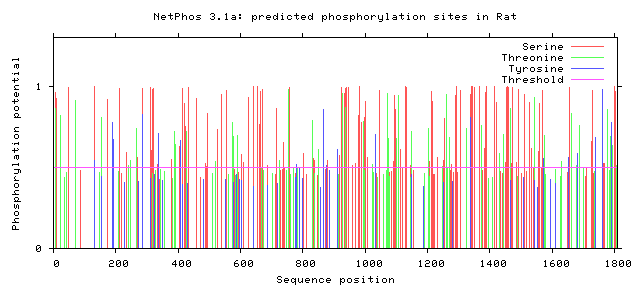
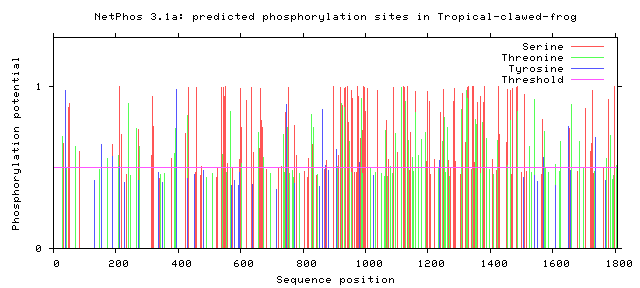
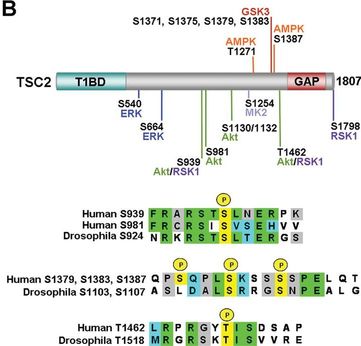
As seen above, there are many potential phosphorylation sites on tuberin protein. By comparing these phosphorylation sites across multiple species, it is possible to identify important sites because these are most likely to be conserved. A sample of this analysis is provided below [2]. It is important to study these sites because they may indicate regulation of or by other proteins. Tuberin, in particular, regulates its interaction with its partner, hamartin, by phosphorylation, in addition to upstream and downstream activities [2][3].
|
References
[1] Hunter, T. (2012). Why nature chose phosphate to modify proteins. Philos Trans R Soc Lond B Biol Sci. 367(1602):2513-6. http://doi.org/10.1098/rstb.2012.0013
[2] Park, S., Chapuis, N., Tamburini, J., Bardet, V., Cornillet-Lefebvre, P., Willems, L., et al. (2010). Role of the PI3K/AKT and mTOR signaling pathways in acute myeloid leukemia. Haematologica, 95(5), 819-828. http://doi.org/10.3324/haematol.2009.013797
[3] Aicher, L. D., Campbell, J. S., & Yeung, R. S. (2001). Tuberin Phosphorylation Regulates Its Interaction with Hamartin. The Journal of Biological Chemistry 276(24), 21017-21021. http://doi.org/10.1074/jbc.C100136200
Image Refererences
Fig 1: http://www.rockland-inc.com/uploadedImages/ProductsStatic/post-translational-modifications-ptm.gif
Fig 2: [2]